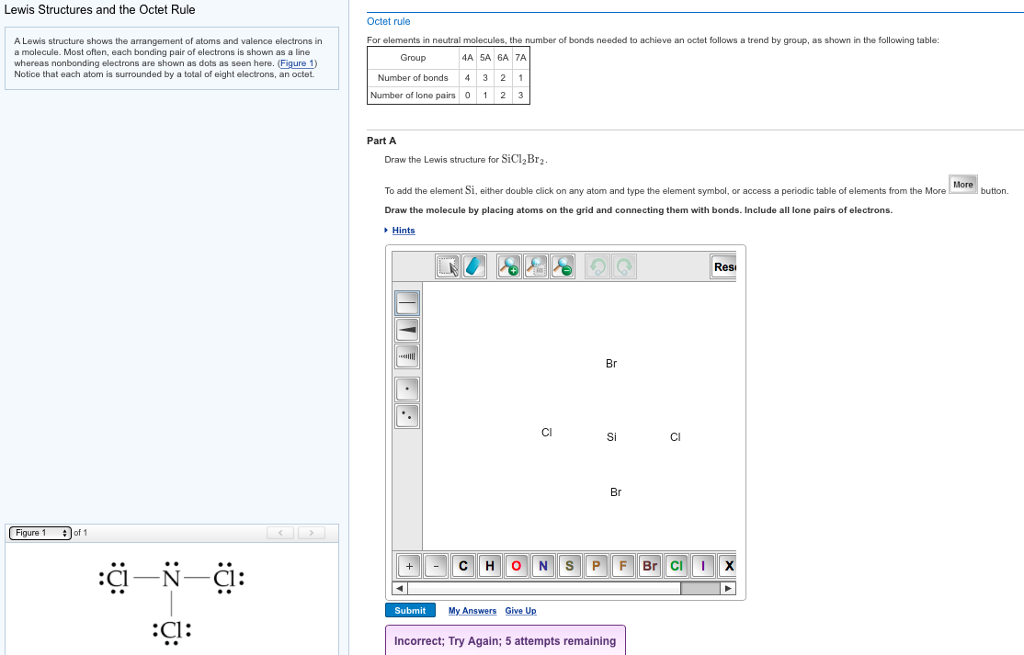
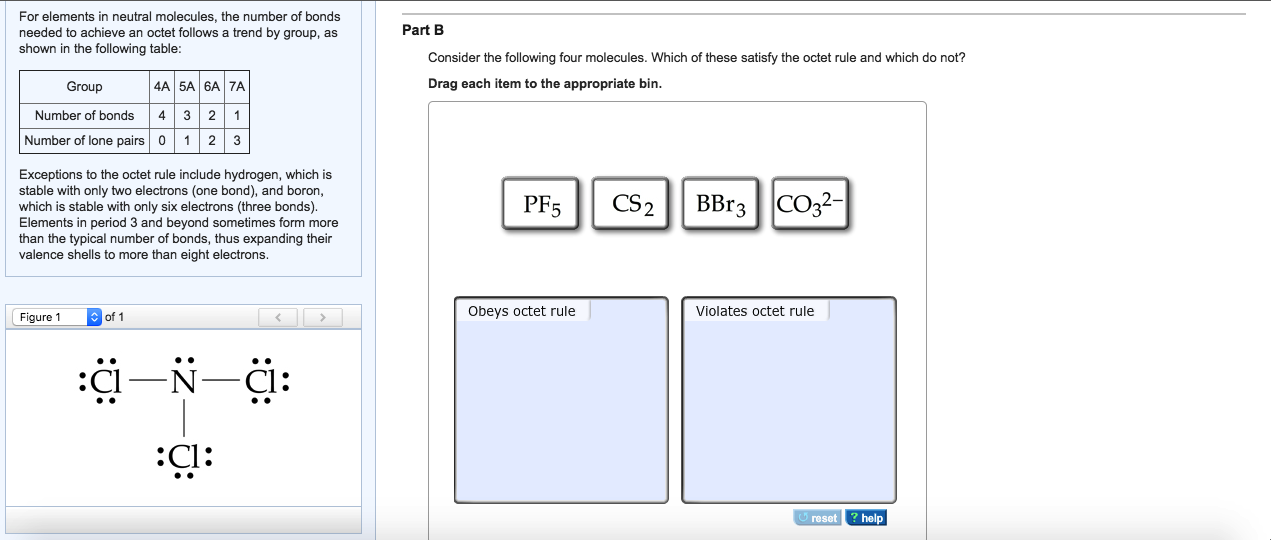
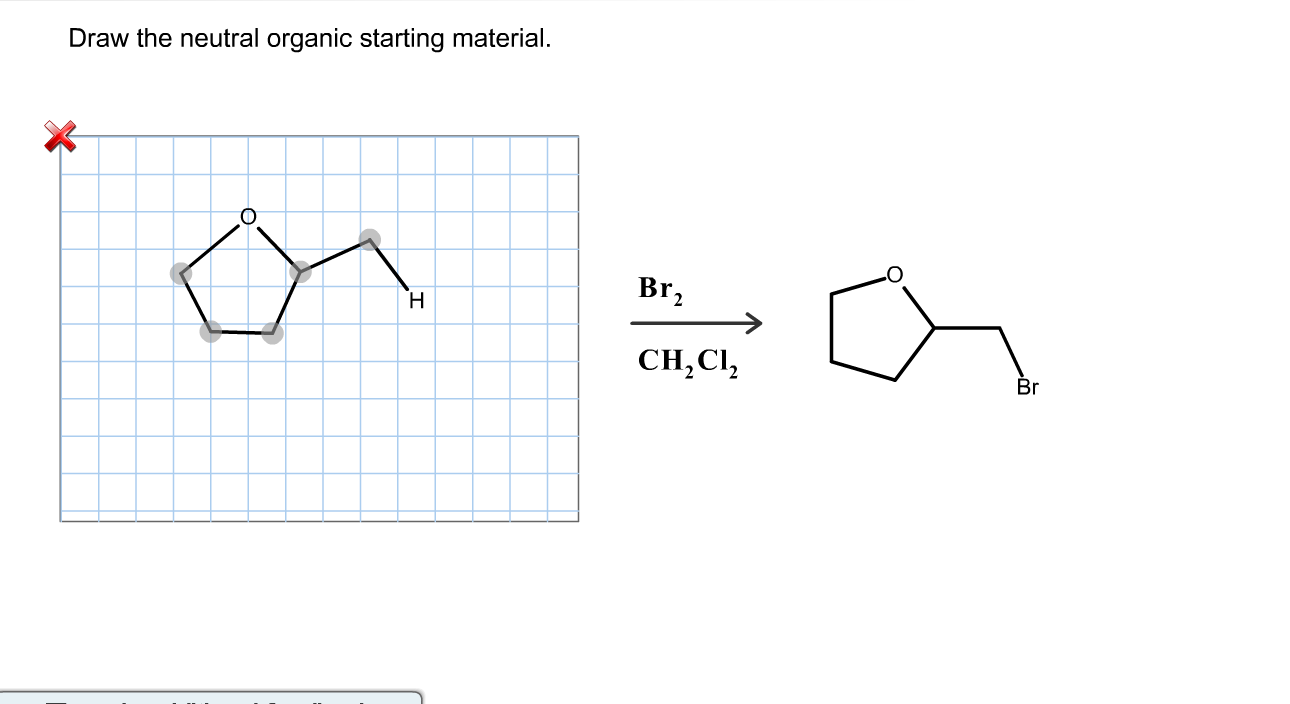
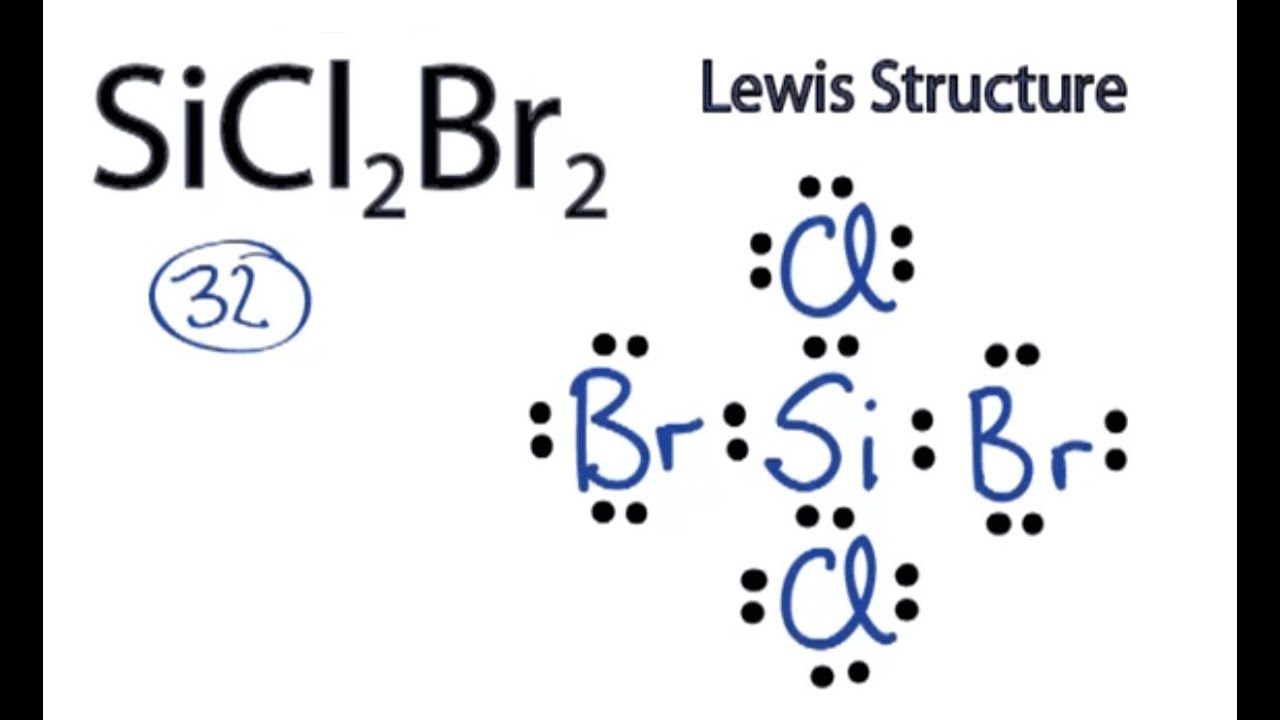Draw The Lewis Structure For Sicl2Br2
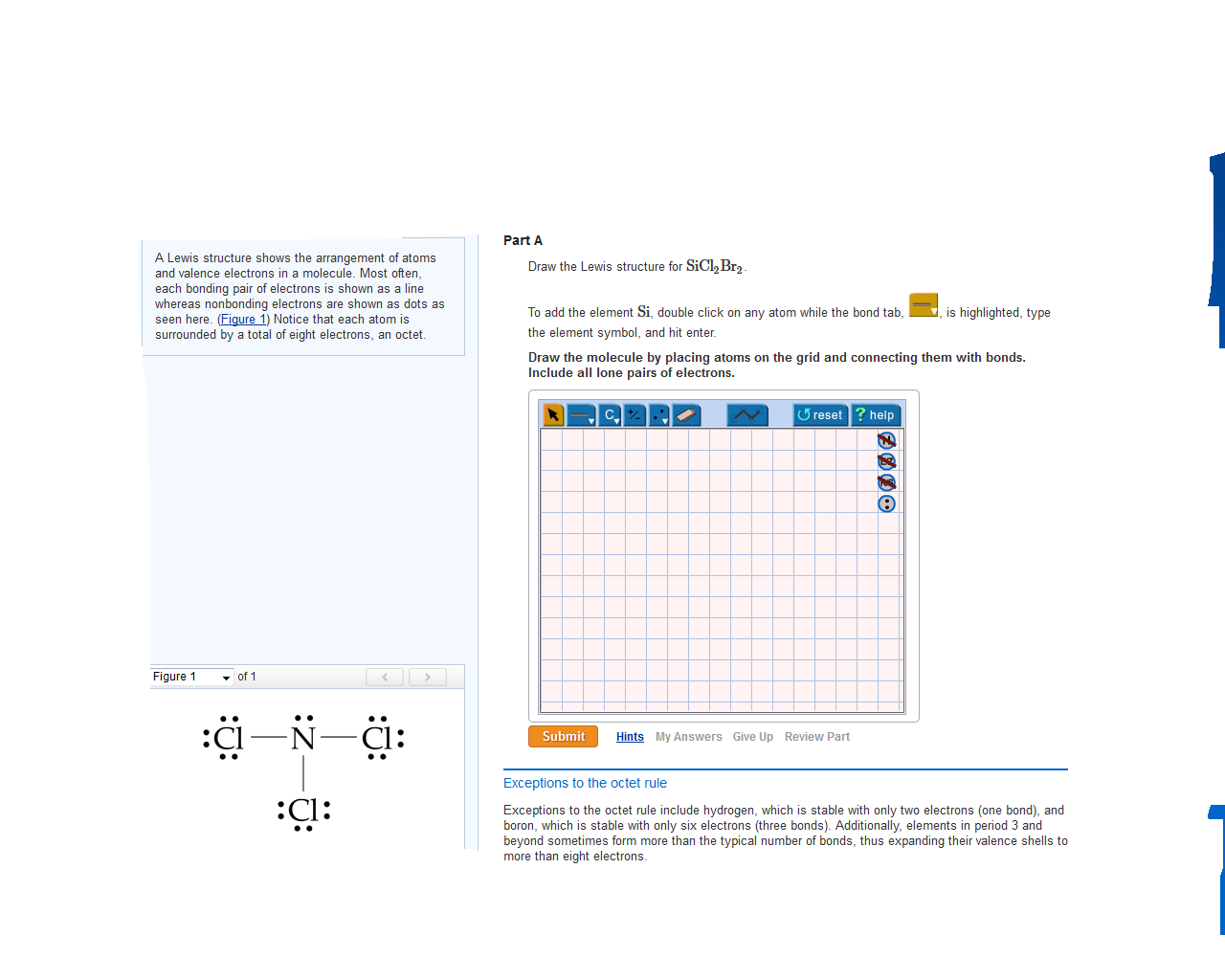
Draw The Lewis Structure For Sicl2Br2 - Web in this article, “sicl2br2 lewis structure”, lewis structure drawing, hybridization, shape, formal charge calculation with some detailed explanations are discussed briefly. To complete the structure, fill the octets of the cl and br atoms with the remaining 24 electrons. #1 draw skeleton #2 show chemical bond #3 mark lone pairs #4 calculate formal charge and check stability (if octet is already completed on central atom) Figure out how many electrons the molecule must have, based on the number of valence electrons in each. Include all lone pairs of electrons. Draw the molecule by placing atoms on the grid and connecting them with bonds. Web steps to draw the lewis structure of sicl2br2. Lone pairs, unpaired electrons, and. Therefore, the total number of valence electrons in sicl2br2 is: Firstly, silicon (si) is the least electronegative and is chosen as the central atom and the others are arranged around it. To complete the structure, fill the octets of the cl and br atoms with the remaining 24 electrons. Web steps of drawing sicl2br2 lewis structure step 1: To add the element si, either double click on any atom and type the element symbol, or access a periodic table of elements from the more button. Include all lone pairs of electrons. Valence electronic structures can be visualized by drawing lewis symbols (for atoms and monatomic ions) and lewis structures (for molecules and polyatomic ions). Include all lone pairs of electrons. Draw the molecule by placing atoms on the grid and connecting them with bonds. Here, the given molecule is sicl2br2. Lewis structures are very similar to electron dot diagrams except for the fact that shared electrons between atoms are shown as lines. Web this widget gets the lewis structure of chemical compounds. Web how do you draw a lewis structure for sicl_2br_2? Draw the molecule by placing atoms on the grid and connecting them with bonds. #1 draw a rough sketch of the structure #2 next, indicate lone pairs on the atoms #3 indicate formal charges on the atoms, if necessary Figure out how many electrons the molecule must have, based on. Lewis structures are very similar to electron dot diagrams except for the fact that shared electrons between atoms are shown as lines. Therefore, the total number of valence electrons in sicl2br2 is: View available hint(s) 0 do q? Draw the molecule by placing atoms on the grid and connecting them with bonds. Web in this article, “sicl2br2 lewis structure”, lewis. Web steps of drawing sicl2br2 lewis structure step 1: Draw the molecule by placing atoms on the grid and connecting them with bonds. Count the total number of valence shell electrons on the compound. Therefore, the total number of valence electrons in sicl2br2 is: Valence electronic structures can be visualized by drawing lewis symbols (for atoms and monatomic ions) and. Web draw the lewis structure for sicl, br2. To add the element si, either double click on any atom and type the element symbol, or access a periodic table of elements from the more button. Lone pairs, unpaired electrons, and. Figure out how many electrons the molecule must have, based on the number of valence electrons in each. Lewis structures. Find more chemistry widgets in wolfram|alpha. Sicl 2 br 2 is a tetrahedral compound with sp 3 hybridization. To add the element si, either double click on any atom and type the element symbol, or access a periodic table of elements from the more more button button. Determine the total number of valence electrons in the molecule. Draw the molecule. Web drawing lewis structures for molecules with one central atom: Lone pairs of electrons themselves are usually represented by dots around the atoms. Include all lone pairs of electrons. Determine the total number of valence electrons in the molecule. Web the lewis structure for the molecule sicl2br2 is drawn by following a set of guidelines. Sicl 2 br 2 is a tetrahedral compound with sp 3 hybridization. Choose a suitable central atom for the compound. Find the total valence electrons in sicl2br2 molecule. Firstly, silicon (si) is the least electronegative and is chosen as the central atom and the others are arranged around it. Include all lone pairs of electrons. View available hint(s) 0 do q? Web to properly draw the sicl 2 br 2 lewis structure, follow these steps: Find the total valence electrons in sicl2br2 molecule. It also provides the molecular geometry and bond angle for sicl2br2 as well. Draw the molecule by placing atoms on the grid and connecting them with bonds. It also provides the molecular geometry and bond angle for sicl2br2 as well. Include all lone pairs of electrons. Therefore, the total number of valence electrons in sicl2br2 is: Draw the molecule by placing atoms on the grid and connecting them with bonds. Here’s the best way to solve it. Web this video shows you how to draw the lewis dot structure for sicl2br2. It also provides the molecular geometry and bond angle for sicl2br2 as well. Include all lone pairs of electrons. Lewis structures are very similar to electron dot diagrams except for the fact that shared electrons between atoms are shown as lines. In order to draw the. Web sicl2br2 is a chemical formula for dibromo dichloro silane. Web overall, the lewis structure for sicl2br2 can be drawn as: The following procedure will give you the correct lewis structure for any molecule or polyatomic ion that has one central atom. Web drawing lewis structures for molecules with one central atom: Draw the molecule by placing atoms on the grid and connecting them with bonds. Figure out how many electrons the molecule must have, based on the number of valence electrons in each. Here’s the best way to solve it. Web steps to draw the lewis structure of sicl2br2. Choose a suitable central atom for the compound. Determine the total number of valence electrons in the molecule. Web 6 steps to draw the lewis structure of sicl2br2 step #1: To add the element si, either double click on any atom and type the element symbol, or access a periodic table of elements from the more more button button. #1 draw skeleton #2 show chemical bond #3 mark lone pairs #4 calculate formal charge and check stability (if octet is already completed on central atom) Web this video shows you how to draw the lewis dot structure for sicl2br2. Therefore, the total number of valence electrons in sicl2br2 is: Web draw the lewis structure for sicl, br2.lewis structure for sicl2br2
Draw the Lewis structure for SiCl2Br2. HomeworkLib
lewis structure for sicl2br2
Sicl2br2 Lewis Structure How To Draw The Lewis Struct vrogue.co
Draw the Lewis Structure for Sicl2br2.
SiCl2Br2 Lewis Structure How to Draw the Lewis Structure for SiCl2Br2
SiCl2Br2 Lewis Structure How to Draw the Lewis Structure for SiCl2Br2
Draw The Lewis Structure For Sicl2br2
Sicl2br2 Lewis Structure How To Draw The Lewis Structure
lewis structure for sicl2br2
Valence Electronic Structures Can Be Visualized By Drawing Lewis Symbols (For Atoms And Monatomic Ions) And Lewis Structures (For Molecules And Polyatomic Ions).
Here, The Given Molecule Is Sicl2Br2.
Web Steps Of Drawing Sicl2Br2 Lewis Structure Step 1:
Lone Pairs Of Electrons Themselves Are Usually Represented By Dots Around The Atoms.
Related Post: