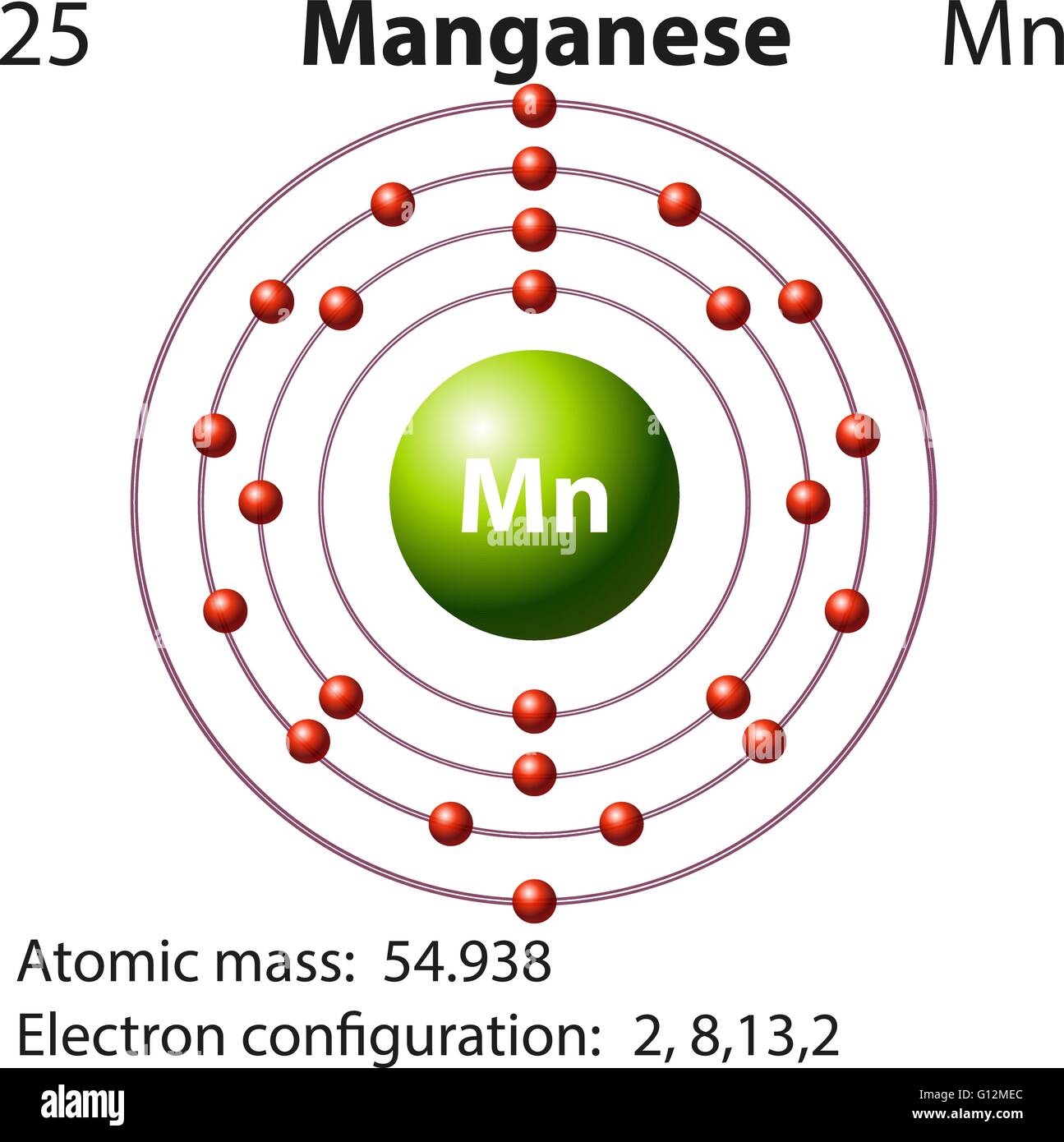
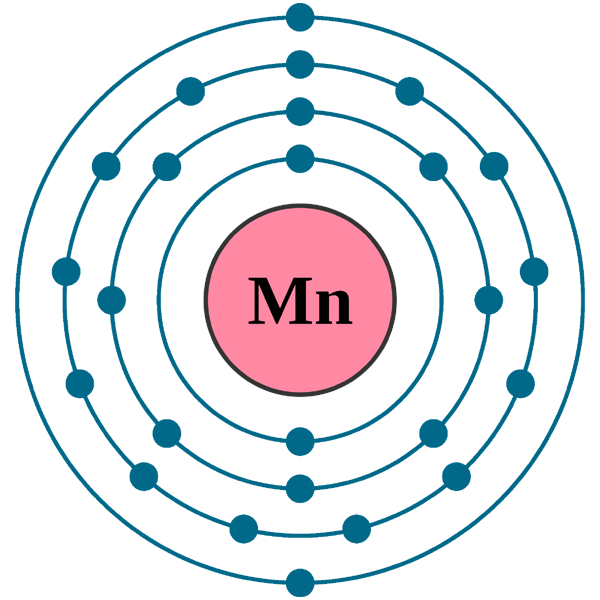
Draw The Electron Configuration For A Neutral Atom Of Manganese
Draw The Electron Configuration For A Neutral Atom Of Manganese - Web the electron configuration of a neutral atom of manganese (mn) is obtained by placing the electrons in the lowest energy levels first, moving up to higher energy. Mn has an atomic number of 25. We first need to find the number of. Web the electron configuration of mn is 1s^2 2s^2 2p^6 3s^2 3p^6 4s^2 3d^5. Web valence electrons are found on the outer shell of an atom and bond. Web using figure \(\pageindex{2}\) as your guide, write the electron configuration of a neutral phosphorus atom. Web to write the configuration for the manganese ions, first we need to write the electron configuration for just manganese (mn). Web find the full electronic configuration and valence electrons of any periodic element using this electron configuration calculator. The atomic number of p is 15. Your solution’s ready to go! Web the electron configuration of mn is 1s^2 2s^2 2p^6 3s^2 3p^6 4s^2 3d^5. Web the electron configuration of an element is the arrangement of its electrons in its atomic orbitals. For most atoms, the magic number is 8 electrons to make a compound stable. #1s^2, 2 s^2, 2p^6, 3s^2, 3p^4#. Web electron configuration chart of all elements is mentioned in the table below. By knowing the electron configuration of an element, we can predict and. The noble gas prior to the element manganese is argon which has 18. [ar]4s^2 3d^5 this is the abbreviated electron configuration of manganese. Web find the full electronic configuration and valence electrons of any periodic element using this electron configuration calculator. Web using figure \(\pageindex{2}\) as your guide, write the electron configuration of a neutral phosphorus atom. Draw the electron configuration for a neutral atom of manganese energy. Web the electron configuration of an element is the arrangement of its electrons in its atomic orbitals. Your solution’s ready to go! By knowing the electron configuration of an element, we can predict and. Web determine the electron configuration of ions. Web the electron configuration of mn is 1s^2 2s^2 2p^6 3s^2 3p^6 4s^2 3d^5. Web an electrically neutral atom has the following electron configuration: What is the name of this atom? Web a quick glance at the periodic table tells me z=25.and we merely follow the aufbau principle. mn, z=25, 1s^ (2)2s^ (2)2p^ (6)3s^ (2)3p^ (6)4s^ (2)3d^ (5). The noble. In an electroneutral atom, the number of the. Web to write the configuration for the manganese ions, first we need to write the electron configuration for just manganese (mn). Elements in group 1 have one. We first need to find the number of. Draw the electron configuration for a neutral atom of manganese energy. Web to write the electron configuration of manganese (m n) \ce{(mn)} (mn) we have to know the number of electrons in its atom. [ar]4s^2 3d^5 this is the abbreviated electron configuration of manganese. The shorthand electron configuration (or noble gas configuration) as well as. Draw the electron configuration for a neutral atom of manganese energy. In the ground state, mn. Web determine the electron configuration of ions. Determine the atomic number of manganese from the periodic table: Web the electron configuration of mn is 1s^2 2s^2 2p^6 3s^2 3p^6 4s^2 3d^5. For most atoms, the magic number is 8 electrons to make a compound stable. The noble gas prior to the element manganese is argon which has 18. Web determine the electron configuration of ions. In the ground state, mn has five unpaired electrons in its 3d sublevel, which gives it a magnetic. #1s^2, 2 s^2, 2p^6, 3s^2, 3p^4#. Web in a neutral atom, the number of electrons is equal to the number of protons in the atom i.e., the atomic number of the element. An atom has. Web to write the configuration for the manganese ions, first we need to write the electron configuration for just manganese (mn). Hence the number of electrons in. An atom has a valence shell electron. We first need to find the number of. Mn has an atomic number of 25. Web the electron configuration of mn is 1s^2 2s^2 2p^6 3s^2 3p^6 4s^2 3d^5. The shorthand electron configuration (or noble gas configuration) as well as. Web to write the electron configuration of manganese (m n) \ce{(mn)} (mn) we have to know the number of electrons in its atom. In an electroneutral atom, the number of the. Determine the atomic number. The noble gas prior to the element manganese is argon which has 18. Web a quick glance at the periodic table tells me z=25.and we merely follow the aufbau principle. mn, z=25, 1s^ (2)2s^ (2)2p^ (6)3s^ (2)3p^ (6)4s^ (2)3d^ (5). Web the electron configuration of an element is the arrangement of its electrons in its atomic orbitals. Web the electron. #1s^2, 2 s^2, 2p^6, 3s^2, 3p^4#. The shorthand electron configuration (or noble gas configuration) as well as. In an electroneutral atom, the number of the. Web electron configuration chart of all elements is mentioned in the table below. Web the electron configuration of mn is 1s^2 2s^2 2p^6 3s^2 3p^6 4s^2 3d^5. Web valence electrons are found on the outer shell of an atom and bond. [ar]4s^2 3d^5 this is the abbreviated electron configuration of manganese. The atomic number of p is 15. Web the electron configuration of an element is the arrangement of its electrons in its atomic orbitals. Web determine the electron configuration of ions. Web to write the electron configuration of manganese (m n) \ce{(mn)} (mn) we have to know the number of electrons in its atom. Web the electron configuration of mn is 1s^2 2s^2 2p^6 3s^2 3p^6 4s^2 3d^5. Elements in group 1 have one. Draw the electron configuration for a neutral atom of manganese energy. Web an electrically neutral atom has the following electron configuration: By knowing the electron configuration of an element, we can predict and. An atom has a valence shell electron. Web to write the configuration for the manganese ions, first we need to write the electron configuration for just manganese (mn). Web the electron configuration of a neutral atom of manganese (mn) is obtained by placing the electrons in the lowest energy levels first, moving up to higher energy. In an electroneutral atom, the number of the. Web electron configuration chart of all elements is mentioned in the table below.Manganese Atom Science Notes and Projects
Manganese Electron Configuration Manganese Orbital Diagram Insight
Draw the electron configuration for a neutral atom of mangan Quizlet
Draw The Electron Configuration For A Neutral Atom Of Manganese
SOLVED Draw the electron configuration for a neutral atom of manganese
Symbol and electron diagram for Manganese illustration Stock Vector
What is the electron configuration for a neutral atom of manganese?
Electron Configuration of Manganesse Mn Lesson YouTube
Manganese electron configuration Newton Desk
Draw The Electron Configuration For A Neutral Atom Of Manganese
In The Ground State, Mn Has Five Unpaired Electrons In Its 3D Sublevel, Which Gives It A Magnetic.
The Shorthand Electron Configuration (Or Noble Gas Configuration) As Well As.
Mn Has An Atomic Number Of 25.
What Is The Name Of This Atom?
Related Post: